
Technology
A Delivery System Modeled on Nature
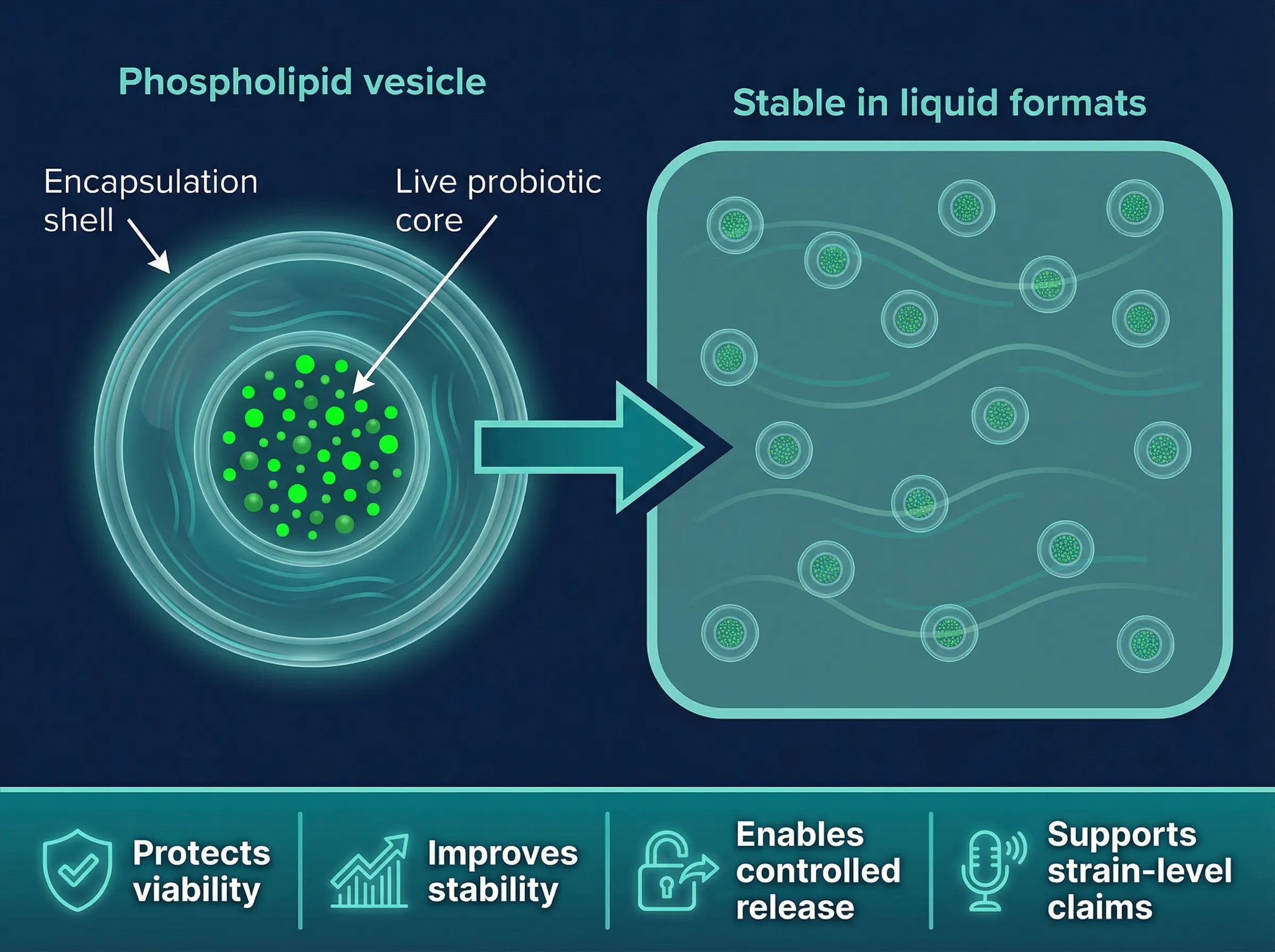
Our patented encapsulation wraps live probiotics in a phospholipid bilayer -- the same protective structure found in every human cell membrane. The result: shelf-stable viability validated to 40°C accelerated conditions with triggered release at skin temperature.
The Mechanism
Phospholipid Bilayer Vesicle Encapsulation
Dakota's core technology encapsulates live probiotic bacteria within a concentric phospholipid bilayer vesicle — composed of FDA-GRAS phospholipids identical to those in human cell membranes. This thermodynamically stable barrier protects probiotics from oxygen, moisture, preservatives, and environmental degradation, eliminating cold chain dependencies entirely.
Stage 1
Before Application
Live probiotics sealed within a concentric phospholipid bilayer vesicle — stable through manufacturing, shipping, and shelf storage.
Stage 2
Upon Skin Contact
Body heat triggers a phase transition in the bilayer. Skin lipids begin exchanging with vesicle phospholipids.
Stage 3
Fusion and Release
The bilayer merges with the skin membrane. Mechanical shear and enzymatic activity complete the opening, releasing viable probiotics.
Stage 4
Post-Release
Metabolically active probiotics colonize the skin surface. Biocompatible bilayer fragments support adhesion and biofilm formation.
Scroll to explore
Stability Data
Proven Shelf Stability
At 25°C after extended storage
At 40°C accelerated conditions
Real-time shelf life validated
Triggered delivery at skin temperature

Delivery Pathways
Multi-Trigger Release System
Upon contact with the target environment, the phospholipid bilayer responds to a convergence of thermal, enzymatic, and mechanical triggers — releasing viable, metabolically active probiotics exactly where they are needed. This multi-pathway release mechanism ensures precise delivery across diverse application formats.
Clinical Outcomes
In-Vivo Study Results
Independent clinical studies demonstrating therapeutic outcomes of live probiotic delivery through our encapsulation platform.
Dermatitis Clinical Outcomes
14 days of topical live probiotics
in dermatitis symptoms (dermatologist score from 1.0 to 0)
in skin hydration (score 3.0 to 4.0)
Independent, validated clinical study performed by Sequential Bio
Results from independent, validated clinical study performed by Sequential Bio. Individual results may vary.
Acne Clinical Outcomes
14 days of topical live probiotics
in acne symptoms (score 1.0 to 0.5)
signs of inflammation (score 4.0 to 3.0)
Independent, validated clinical study performed by Sequential Bio
Results from independent, validated clinical study performed by Sequential Bio. Individual results may vary.
Chemical Peel Recovery
14-day post-procedure assessment
Probiotic treatment group showed increased total bacterial DNA vs. control by day 14
Independent, validated clinical study performed by Sequential Bio
Results from independent, validated clinical study performed by Sequential Bio. Individual results may vary.
All clinical studies referenced above were performed independently by Sequential Bio. Results represent study outcomes and individual results may vary. These statements have not been evaluated by the FDA.
Partner With a Clinically Validated Platform
Dakota's technology is backed by independent clinical data and protected by a growing patent portfolio.
View IP Portfolio